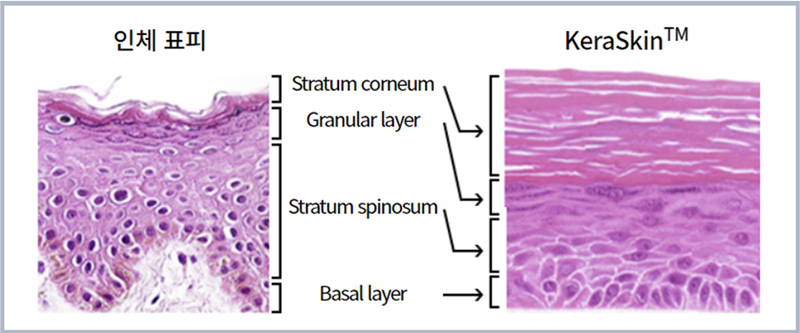
On the right is KeraSkin, a 3D human skin model developed by the Korea Center for the Validation of Alternative Methods created from epidermal keratinocytes from human skin tissue. (Screen capture from center's official website)
By Hong Angie
The reliability of human skin models made in Korea has gained recognition abroad.
The National Institute of Food and Drug Safety Evaluation, an affiliate of the Ministry of Food and Drug Safety, on April 27 said alternative method of animal testing for a reconstructed human epidermis phototoxicity test (KeraSkin) was adopted as the 38th Test Guideline (TG 498) of the Organisation for Economic Co-operation and Development (OECD).
Developed by the Korean Center for the Validation of Alternative Methods, the testing method is a homegrown technology that breaks away from reliance on foreign know-how. It uses a human skin model to assess the phototoxicity of cosmetic ingredients and other substances as an innovative alternative to sacrificing animals in laboratory tests.
The adoption of the method was confirmed on April 23 at the meeting of the OECD Working Party of the National Coordinator of the Test Guidelines Programme. KeraSkin is the second human skin model listed among the OECD's 38 member countries, following one from the U.S.
The model has high commercial value. The domestic cosmetics industry can slash testing costs and shipping times incurred by high-priced foreign models as well as accelerate its advance into overseas markets through this development.
The ministry said, "We will strengthen institutional support to allow the stable settlement of alternative methods of animal testing in cosmetics policy."
shong9412@korea.kr